|
11/21/2023 0 Comments Formal charge calculation practice 
Simply calculate which atom carries charge. (Video below included in this tutorial.) Video 4 – Formal Charge Calculation Shortcut This detailed written tutorial will teach you my quick shortcut and is followed by lots of practice questions. This video breaks down sp2 hybridization and sp fused orbitals using carbon and other atoms to help you understand hybrids, geometry, and bond angles.įormal charge calculations are taught as a complicated and confusing process. Sp2 and sp hybrids give organic compounds unique shapes and reacting abilities. 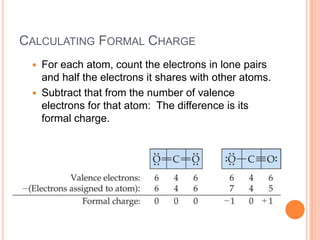
Video 3 – sp2 and sp Hybridization and Bond Angles For more on sp3 hybridization for carbon and other atoms, geometry and bond angle, see my video below. This ability of binding 4 different atoms for a 3-D structure is unique to organic compounds. Video 2 – sp3 Hybridization and Bond AnglesĬarbon must have 4 bonds. Click here for an in-depth overview (+ videos) of sp³, sp² and sp Hybridization, Geometry and Bond Angles. These are crucial to understanding reactions. Learning these concepts will help you recognize strong/weak bonds and bonds that can or can’t be broken. Video 1 – Ionic, Polar Covalent and Non-Polar Covalent Bonding Have trouble classifying primary, secondary, and tertiary carbon atoms? You can't get far without this skill, and this trick makes its simple! I'll also cover molecules containing oxygen bonds, nitrogen, and more. Pre-Basics Video – Skeletal StructuresĬonfused by those sticks and lines masquerading as molecules? In this video, you’ll learn how to identify molecules when written in line structure. 
Worried you're behind? Go back to the ‘Gen Chem Review for Orgo' series first and test your knowledge with the Gen Chem for Orgo Quiz!įor a quick review, stop and watch my ‘ General Chemistry Review for Organic Chemistry Students‘ video. That's why this video series is designed to teach you these very foundation topics that were touched on in general chemistry. So now that we know the formal charge formula, click on to the next video on, Let's put it to practice with the given example.Success in organic chemistry requires a strong foundation and understanding of the basics. This is the sum off all formal charges within a compound. We're going to say remember Valence electrons equals the group number off that element, and when we're talking about non bonding electrons, realized that we're gonna count them individually with formal charge when we find the formal charge of every element within a compound, we can calculate what's called the Net charge. We'd say formal charge equals the valence electrons off that element that we're examining minus the bonds that it is making, plus the non bonding electrons that it possesses. Bonding electrons are the electrons that do participate in bonding with other elements, and non bonding electrons are the electrons that don't participate and bonding with other elements with formal charge. We assume that all atoms that are sharing electrons between each other they share them equally now with formal charge were introduced to terms such as bonding, electrons versus non bonding electrons. 
But when it comes to formal charge, we don't take that into consideration. In reality, sometimes an element that's more electro negative will hold on to that electron within the electrons within the bond a little bit more tightly around themselves, which inflates or makes the electron clouds larger. Now formal charge represents the charge given toe elements when assuming electrons are shared equally, regardless off electoral negativity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
